Explore our network of country and industry based websites to access localized information, product offerings, and business services across our group.
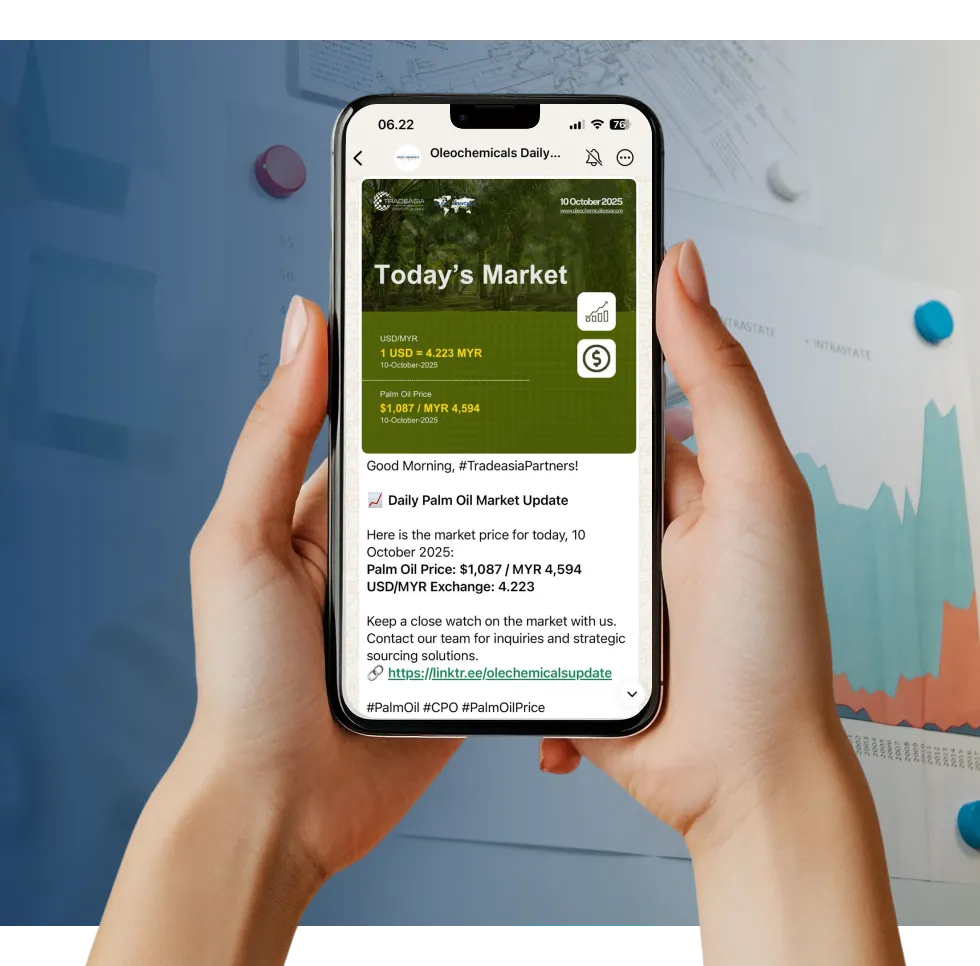
Access reliable chemical market information through our update channels.
Real-time Updates
Daily Updates
Log in to start sending quotation requests for any product.
Don't have an account? Sign Up Here
Log in now to access technical product documents from our product range.
Don't have an account? Sign Up Here
Home All Products Phthalic Anhydride (99,8%) - Taiwan
|
IUPAC Name |
: 2-Benzofuran-1,3-dione |
|
Cas Number |
: 85-44-9 |
|
HS Code |
: 2917.35.00 |
|
Formula |
: C8H4O3 |
|
Appearance Name |
: Fine White Powder |
|
Common Names |
: Isobenzofuran-1,3-dione, Phthalic anhydride |
|
Packaging |
: Flake Bag: 750kg、600kg、500kg、25kg , Palletized |
For more detailed information including pricing, customization, and shipping:
Description:
Phthalic anhydride is the organic compound with the formula C6H4(CO)2O, , serves as the anhydride of phthalic acid and represents a crucial commercial form of the acid. It stands as a pioneering example of the widespread utilization of a dicarboxylic acid anhydride, playing a vital role in industrial chemistry. Specifically, it plays a key role in the extensive production of plasticizers for plastics, contributing to the estimated global production volume of around 3 million tonnes annually in 2000.
Manufacturing Process:
Discovered by Auguste Laurent in 1836, the contemporary synthesis of phthalic anhydride employs various methods, including the oxidation of naphthalene or ortho-xylene. These processes incorporate vanadium pentoxide (V2O5) as the active oxidant, playing a pivotal role in multiple steps and being regenerated by molecular oxygen. Starting from o-xylene, the oxidation reaction is run at about 320–400 °C and has the following stoichiometry:
C6H4(CH3)2 + 3 O2 → C6H4(CO)2O + 3 H2O
The reaction proceeds with about 70% selectivity. About 10% of maleic anhydride is also produced: C6H4(CH3)2 + 7.5 O2 → C4H2O3 +4 H2O + 4 CO2
Phthalic anhydride and maleic anhydride are recovered by distillation by a series of switch condensers.
The naphthalene route (the Gibbs phthalic anhydride process or the Gibbs–Wohg naphthalene oxidation reaction), a process whose use has declined in compared to the o-xylene route, has the following mechanism:
Phthalic anhydride can also be prepared from phthalic acid by simple dehydration.
Phthalate esters plasticizers
The primary application of phthalic anhydride lies in its role as a precursor to phthalate esters, crucial plasticizers for vinyl chloride. The production of these esters, derived from alcoholysis reactions with phthalic anhydride, reached an annual scale of approximately 6.5×109 kg in the 1980s, showcasing a continuous increase, all originating from phthalic anhydride. Additionally, it finds extensive use in manufacturing polyester resins and has minor applications in alkyd resins for paints and lacquers, specific gums, insect repellents, and urethane polyester polyols.
Precursor to dyestuffs
In the dyestuff production sector, phthalic anhydride plays a significant role, notably contributing to the synthesis of the anthroquinone dye quinizarin through reactions with para-chlorophenol followed by chloride hydrolysis. The well-known compound phenolphthalein is synthesized by condensing phthalic anhydride with two equivalents of phenol under acidic conditions, giving it its name.
Pharmaceuticals
In the pharmaceutical industry, treating phthalic anhydride with cellulose acetate produces cellulose acetate phthalate (CAP), a common excipient used for enteric coatings. CAP has demonstrated antiviral activity, and phthalic anhydride itself is a degradation product of CAP.

